An advanced tool for 3D conformational and configurational analysis using experimental constraints from NMR spectra
Mnova StereoFitter computes the probability of 3D structural configurations and/or conformations, based on various forms of NMR experimental data input. Currently, StereoFitter can accept four distinct types of input in order to calculate the best 3D structure candidate(s): NOEs, RDCs, Js, and chemical shifts.
A license of Mnova NMR (version ≥14.1.1) is required for Mnova StereoFitter!
Select a suitable installer below and load it into Mnova (follow the menu “File/Open”)
Please note it is not available in the ‘Advanced Plugins’ panel until installed
What’s new in Mnova StereoFitter 1.1.6?
The latest version 1.1.6 was validated against Mnova 15.0.1, and these are the main highlights:
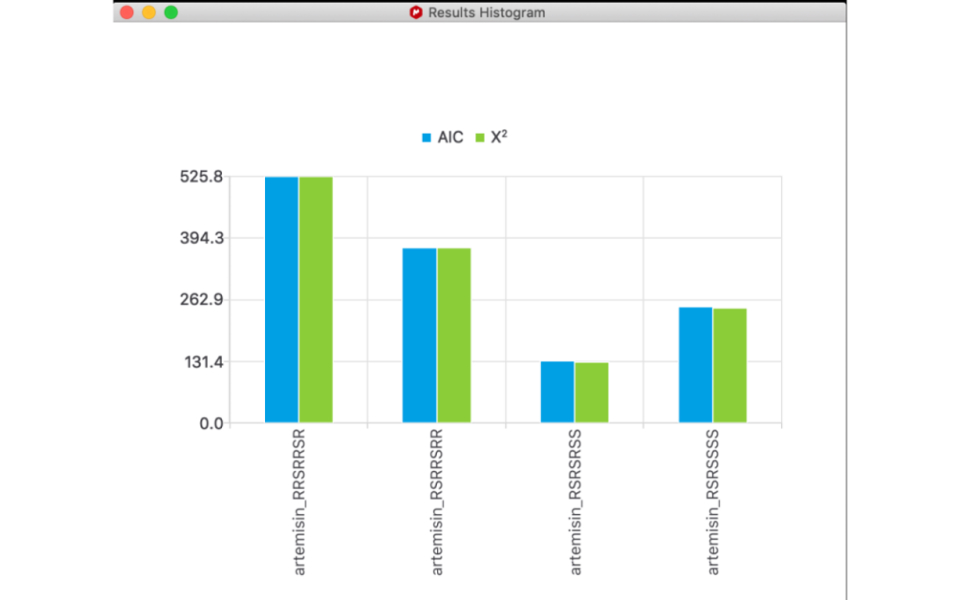
- In the graphical interface, the new normalized “x^2” parameter is now displayed in the results, both in the table and in the bar chart.
Mnova StereoFitter: 45-day free trial
 1. Download
1. Download
Download Mnova and Mnova StereoFitter for Windows or Mac
 2. Installation
2. Installation
Open Mnova and go to ‘Help /Get-Install Licenses’. Select ‘Evaluate’ for a trial
 3. License
3. License
Fill in the form to receive your trial license via e-mail.
IMPORTANT! You must also download an Installer for StereoFitter.
Simply choose a button below and then load it into Mnova.
Mnova StereoFitter
Features
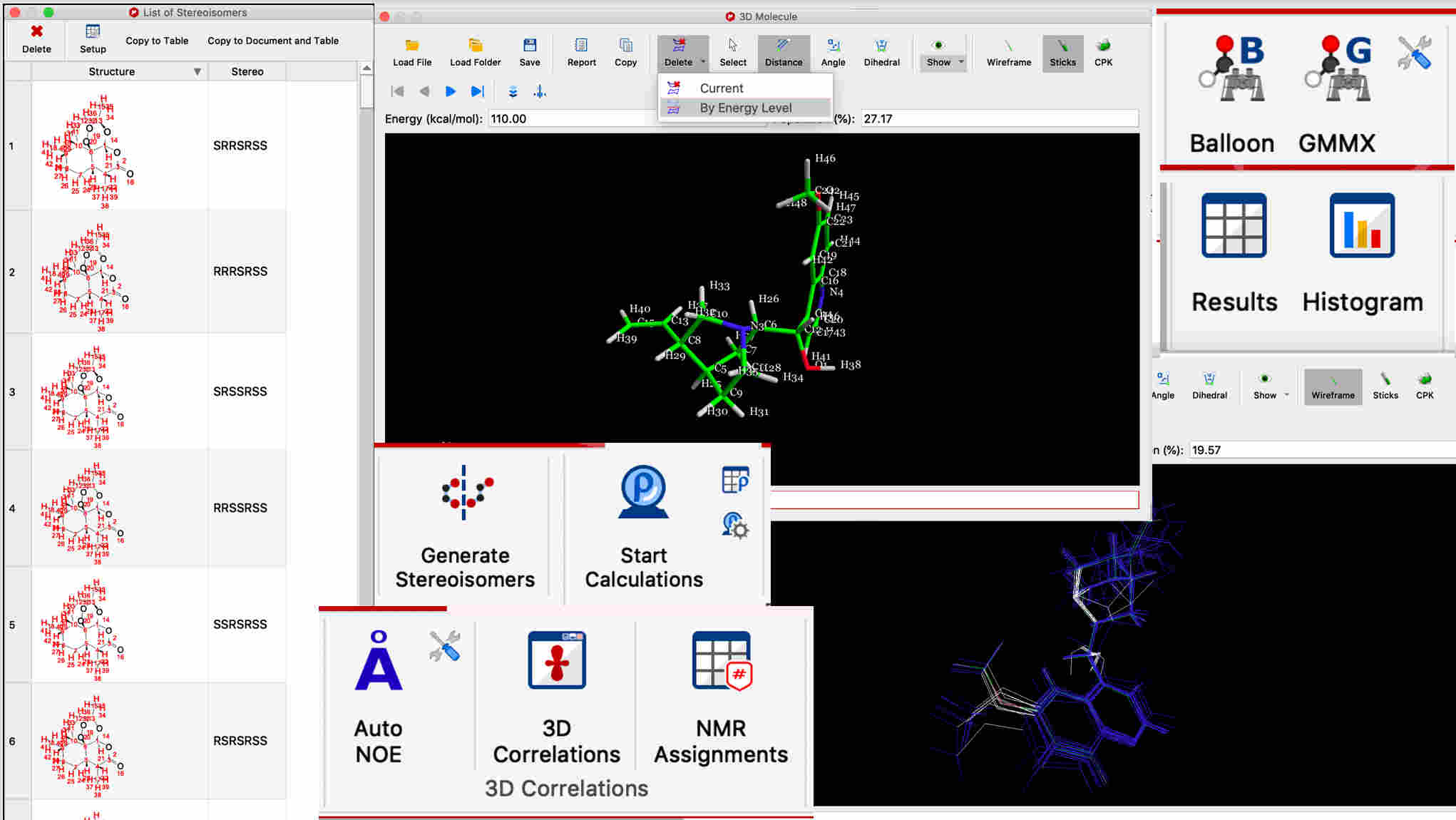
- 3D conformations can be calculated from the 2D structure
- Sets of 3D conformations can be interactively viewed, aligned, overlayed, and filtered by energy level
- Stereoisomers can be computed from any 2D structure with ambiguous stereo centers
- All types of measured constraints can be used in one 3D calculation for more accuracy and precision
For NOEs, distances are easily calculated from measured cross-peak volumes with a single click.
You can find an example in the manual (p.27) of Mnova StereoFitter using NOEs to determine 3D structure from a set of conformers.
For RDCs, automatically calculate RDC values from isotropic and anisotropic spectra.
You can find an example in the manual (p.35) of Mnova StereoFitter using RDCs and predicted chemical shifts to determine 3D structure from a set of stereoisomers.
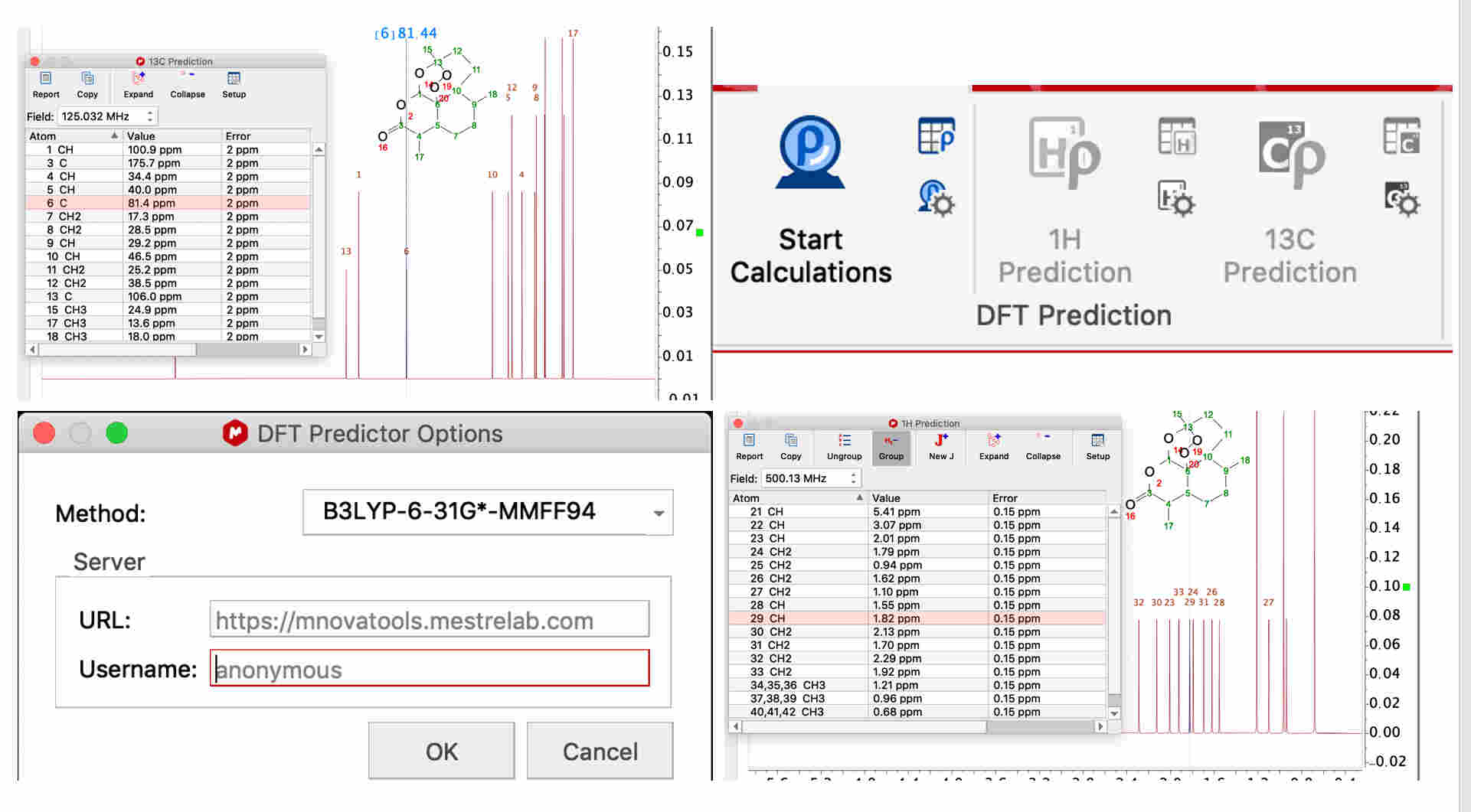
For chemical shift prediction, DFT calculations can be carried out within StereoFitter. A separate DFT Predictor license is required for running these calculations. The DFT Predictions section of the ribbon contains tools to predict 1H or 13C chemical shifts using results from a server-based DFT
program.
You can find more information in the manual (p.24)
Academic, Government & Industrial
Markets
Who should be using Mnova StereoFitter?
- Medicinal chemists or chemists in a Drug Discovery environment
- Computational chemists
- Academic research and development groups
- Analytical chemists dealing with stereochemistry problems
- Other industries with questions about stereochemistry